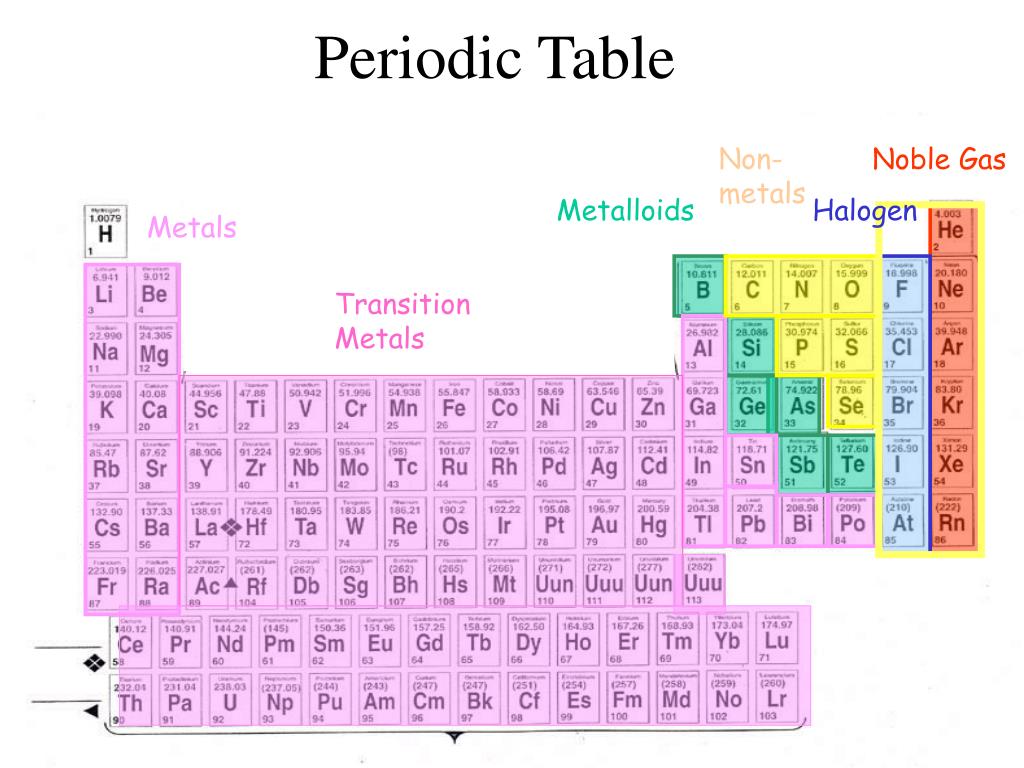
Period 4 elements are quite stable and many of them are very common in earth’s crust or core or both. We can memorize these elements across the periods: Group 12 elements, which have its d subshell completely filled, are also known as post-transition elements.ĭ-block elements and F-block elements show considerable similarities across the periods too. Groups 3 to 11 are also known as transitional metals. D-Block Elementsĭ-Block elements consist of element groups 3 to 12 that correspond to the filling of the d-orbital subshell of the second outermost shell. 
Mnemonic for Group 18: He Never Arrived Ka ra Xero Ru n pe out. The group includes Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and the radioactive Radon (Rn). Normally, they are all odorless and colorless gases with very low chemical reactivity. Group 18 is known as the group of Noble gases, excluding Helium. Mnemonic for Group 17: Fir Cal l kar Bahaa r Aay I Aun ty. It includes Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I), and Astatine (At). Group 17 is known as the group of Halogens. 
Mnemonic for Group 16: Oh! Style Se Tel Polish. It includes Oxygen (O), Sulphur (S), Selenium (Se), Tellurium (Te), and the radioactive element Polonium (Po). Group 16 is known as the group of Chalcogens or Oxygen group. Mnemonic for Group 15: Nahi Pasand Ai se Sa b Bha i. It includes Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), and Bismuth (Bi). Group 15 is known as the group of Pnictogens or Nitrogen group. Mnemonic for Group 14: Chemistry Sir Giv es Sa nki Pro blems. It includes Carbon (C), Silicon (Si), Germanium (Ge), Tin (Sn), and Lead (Pb). Group 14 is known as Carbon group or the group of Crystallogens, Tetragens or Tetrels. It includes Boron (B), Aluminium (Al), Gallium (Ga), Indium (In), and Thallium (Tl). Group 13 is known as Boron group or the group of Icosagens or Triels.  This block consists of non-metals, semi-metals and poor metals. Mnemonic for Group 2: Beta Man ge Car Scoote r Baap rone se Raazi P-Block ElementsĬonsisting of last six groups of the periodic table (Groups 13 to 18), P-block elements have their valence electrons occupying p-orbitals. It includes Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Br), and Radium (Ra). Group 2 is known as alkaline earth metals. Mnemonic for Group 1: LiNa Ki Ruby Cse Friendship hai. It includes Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Ru), Caesium (Cs), and Francium (Fr). The valence electrons of the elements in this block occupy s-orbitals. S-Block ElementsĬonsisting of the first two groups, S-block elements have quite similar physical and chemical properties. • At some places, phonetics have been used to denote a symbols such as ‘c’ could be replaced by ‘k’,’g’ with ‘j’, ‘I’ with ‘ea’ and ‘o’ with ‘u’, to make the sentence easier to remember. While reading the sentence you don’t have to read the letters in bracket. However at the places where the complete symbol could not be included in the sentence, the first letters have been strung together and the second letter is shown in brackets. • The symbols have been highlighted as bold letters in the sentence. • These sentences contain letters denoting symbols of elements in the same order as they occur in a group or period. Key To Reading These Mnemonics Or Hindi Sentence: Here are some fun, interesting and naughty mnemonics in Hindi used by the backbenchers to memorize elements along each group or period: It serves as a great tool for solving chemistry problems.Ī periodic table is divided into groups (columns), where elements with each group behave similarly while bonding with other elements and periods (rows), where elements in one period have same number of electron shells. The Periodic Table provides the names, atomic numbers, symbols and atomic weights of known elements. In this article, you will find Hindi mnemonics – one each for one group – to learn the Periodic Table Mnemonics are easy-to-remember lines or phrases one can use to memorize things that are difficult to learn.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
